Empowering Precision Medicine, One Biospecimen at a Time
At Sona Biosciences, we're not just collecting samples — we're fueling the future of healthcare. Founded in 2023 in Ahmedabad, India's clinical research epicenter, we've rapidly become a cornerstone in the quest for groundbreaking cancer therapies and diagnostics.
Our vast network of 100+ multi-specialty clinical sites spread across India, grants researchers access to a treasure trove of over 100,000 ethically sourced biospecimens and tapestries of patient histories.
But we're more than just numbers. We're a team of passionate experts, blending cutting-edge clinical research capabilities with unwavering ethical standards to deliver the highest quality biospecimens. From the USA to the EU and APAC, we're the trusted partner for researchers big and small, all united by a single mission: to unlock the secrets of human biology and pave the way for a healthier tomorrow.

SONA’s Offerings
In the grand narrative of medical research, we're the plot twist you didn't see coming. Founded in 2023, we burst onto the scene as India's first full-service Biospecimen CRO.
Think of us as the Sherlock Holmes of the biological world – only instead of a magnifying glass, we wield cutting-edge science and an extensive network of clinical sites.
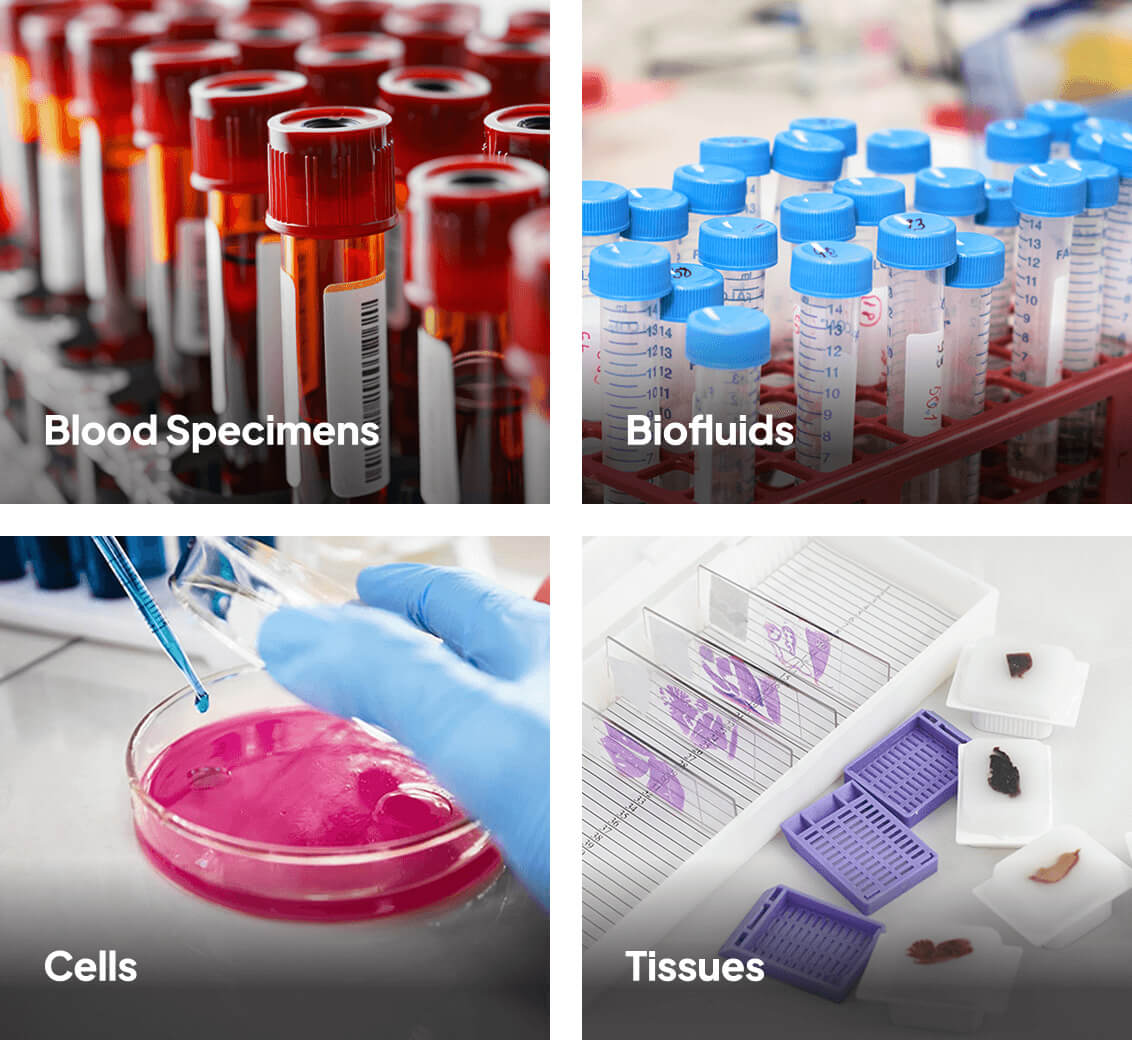
Biospecimens
Lab Services
CDO Services
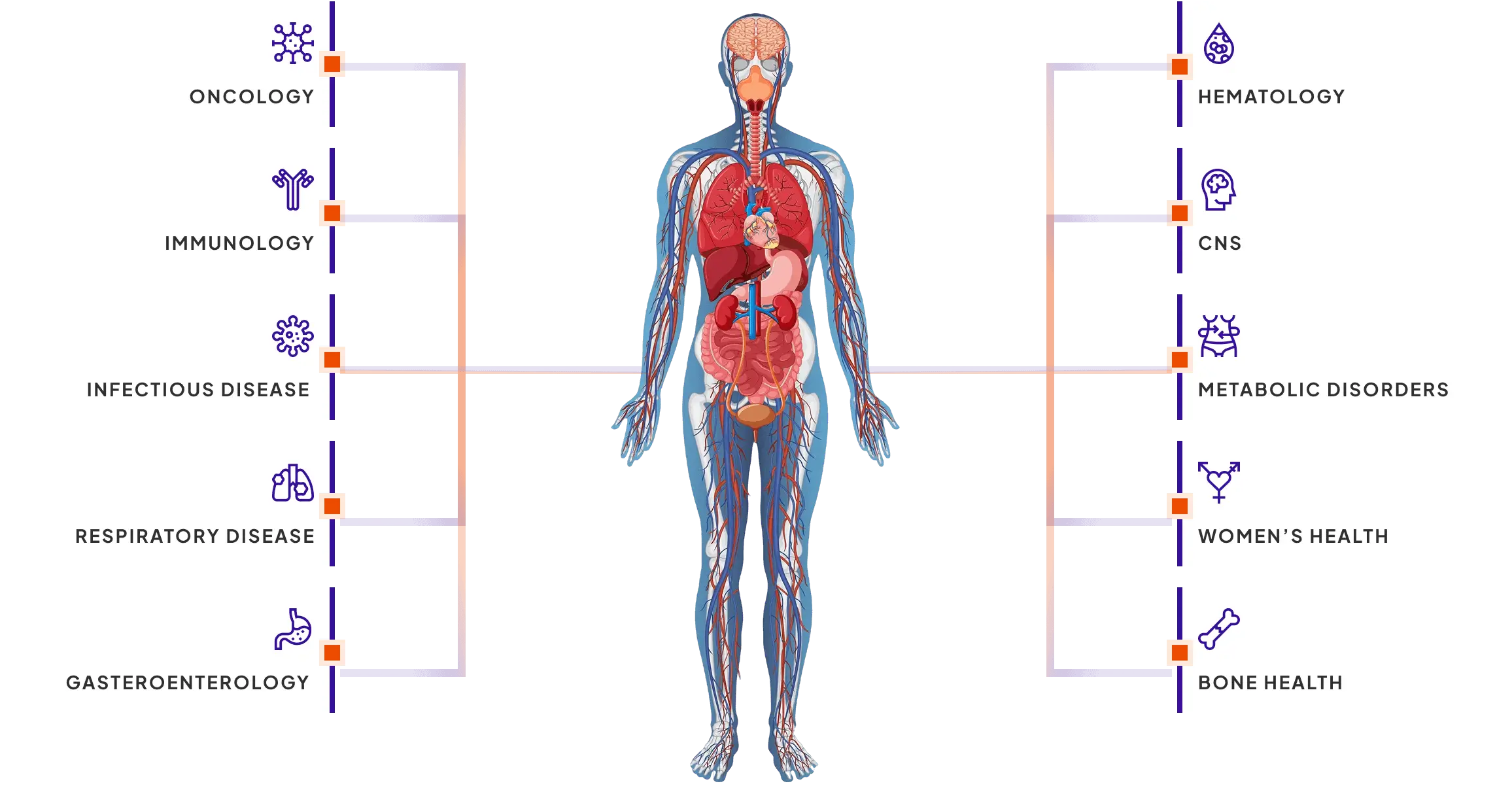
Decipher the Intricacies
In Diverse Diseases
Sona Bioscience offers premium human biospecimens that support
a broad spectrum of disease indications, including

SONA Bioscience’s
Integrity Promise
We take biospecimen sourcing beyond the ordinary! Think of us as the integrity trendsetters, blending ethical practices with a sprinkle of innovation.
We promise quality, consent, and privacy—because doing the right thing should always be in style!
ExploreCommitment to Ethical Biospecimen Sourcing
We prioritize ethical practices in every step of biospecimen collection to ensure integrity and respect for participants.
High-Quality, Consented Samples
Our biospecimens are sourced with appropriate consent, guaranteeing the highest quality for research and clinical applications.
Adherence to IRB-Approved Protocols
We strictly follow Institutional Review Board (IRB) protocols to ensure compliance and ethical standards in all research endeavors.
Robust Patient Data Protection
We implement stringent measures to safeguard patient data, ensuring confidentiality and security throughout the research process.
Reliable and Accurate Data for Research
Our commitment to quality assurance ensures that the data derived from our biospecimens is both reliable and precise, supporting impactful research outcomes.
Our Pan-India Site Network
Powering Diverse Research
Across India, forming the cornerstone of our research capabilities
Corporate
Hospitals
Government
Hospitals
Academic
Institutes
Pathology
Laboratories
Medical
Clinics
Flexible Collection
Capability for both prospective and retrospective biospecimen collections.
Comprehensive Data
Access to well-curated patient data from diverse populations.
Diverse Healthcare Settings
Our network spans corporate hospitals, government hospitals, academic institutes, pathology laboratories & community clinics.
Quality Assurance
Rigorous processes ensure high-quality biospecimens and reliable data across all sites.
Ethics Committee Approval
The network ensures that all clinical sites operate under the supervision of accredited ethics committees to safeguard patient rights and data integrity.
Therapeutic Expertise
Strong connections with oncology and other medical speciality clinical sites.
Customization
Ability to meet specific research requirements, including rare or ethnicity-specific samples.
Geographical Reach
Coverage across multiple regions, offering varied patient population with different cultural and lifestyle differences.
Why is India the
Biospecimen Research Frontier?
India offers unique advantages for biospecimen collection and research
Diverse Patient Pool
Access to a large, genetically diverse population meeting complex inclusion/exclusion criteria.
Cost-Effective
Significant cost and timeline efficiencies compared to other regions.
English Documentation
All site documentation is available in English, facilitating seamless communication.
Regulatory Advantage
Favorable regulatory environment for biomedical research.
ICH GCP Compliance
Sites and staff trained to meet international Good Clinical Practice standards.
Experienced Professionals
Access to qualified, trained, and experienced investigators and site staff.
SONA's Expertise
As a leading Indian CRO, we offer unparalleled local knowledge and connections.
Growing Infrastructure
Expanding clinical trials infrastructure, with numerous active sites, ensures a steady supply of clinical data and biospecimens for global research.
Together, Let’s Take a Stand
Against Debilitating Diseases!
Be a catalyst for change and join our mission for a Healthier Tomorrow - donate your biospecimen or team up as a site partner to explore the complexities of diseases and help researcher develop groundbreaking therapies and diagnostics for patients who need it the most. Together, let’s stir up some science magic!
Power-Innovators
We Serve
Empowering innovators with customized biospecimen solutions for groundbreaking research and development
Biobank
Drug discovery companies
Diagnostic companies
Biospecimen Marketplace
Innovator Biopharama companies
Research-led Universities
Preclinical CROs
Clinical CROs
Bioanalytical CROs