Capabilities
Streamlined process, Unmatched Efficiency, Unrivaled Quality - Your Research, Our Priority.
Feasibility Assessment
A feasibility assessment evaluates a project's practicality and potential success.
Comprehensive Landscape Analysis
We begin with a thorough assessment of the disease landscape, covering prevalence, incidence rates, standard of care practices, patient safety, and cost estimates. Our feasibility studies ensure that we provide realistic estimates aligned with clinical and market demands for your project.
Questionnaire-Based Site Evaluation
We collaborate with participating investigators and clinical sites through a structured questionnaire process. This helps identify potential risks and provides a clear understanding of each site’s infrastructure and capabilities.
In-Depth Site Discussions
Our team conducts detailed site discussions to identify risk areas, evaluate site readiness and ensure a comprehensive understanding of the competitive landscape. We assess the level of commitment from investigators and staff, providing you with accurate insights.
Real-Time Feasibility & Quick Turnaround
With real-time feasibility evaluations, we ensure precise cost estimation and realistic timelines for project execution, offering swift decision making capabilities for your study.

Medical Writing
Protocol, ICF and CRF Design

Medical Writing
Protocol, ICF and CRF Design
Expert Protocol, ICF and CRF Design
Our team of highly experienced medical writers creates documents in compliance with Good Clinical Practice (GCP) and other applicable regulatory standards. With over 100 customized collection protocols drafted, we support clinical study documentation needs, ensuring they meet the highest standards of accuracy and clarity.
Support for Medical Affairs and Commercialization
Beyond protocol development, our writing team supports broader objectives, including publications and medical affairs, ensuring that all project-related documents align with both scientific and commercial goals.
Investigator, Site Identification
and Qualification

Targeted Site Selection and Qualification
We thoroughly evaluate each site, considering therapeutic expertise, patient population diversity and their historical performance in clinical trials. Our selection process is designed to optimize participant recruitment and ensure quality execution.
Regulatory Compliance and Site Auditing
Our team monitors Institutional Review Board (IRB) requirements and performs multiple on-site visits to ensure data integrity, regulatory compliance and alignment with your study objectives. We also conduct quality check on the clinical data and the biosamples to rectify any errors or omissions, ensuring overall quality.
Project Management

Streamlined Communication with a Single Point of Contact
We offer a single-window approach, where a dedicated project manager serves as your primary point of contact. This ensures clear communication and streamlined project execution from start to finish.
Proactive Risk & Timeline Management
Our project management team proactively assesses risks and timelines, ensuring that deliverables are met efficiently. By leveraging our global systems and processes, we ensure accuracy and consistency across all phases of the project.
Vendor Management & Patient Recruitment Strategies
We handle third-party vendor management and implement patient recruitment strategies, keeping you updated on progress through a user-friendly dashboard.
Sourcing Kits and Consumables
Efficient Procurement Solutions
SONA simplifies your procurement process by offering one-stop sourcing for high-quality laboratory kits and consumables. We ensure that all materials meet protocol requirements and are delivered on time and within budget.
Customized Solutions for All Collection Needs
Our bespoke kits are tailored to your specific clinical protocol, ensuring that they include the necessary equipment, packaging, and compliant labeling for easy tracking and accurate sample collection.
Sourcing Kits and Consumables
Labeling and Kit Management

Labeling and Kit Management
Bespoke Kit Design
From blood collection kits to tissue biopsy and genetic testing, our customizable kits meet a wide range of clinical requirements. Each kit is rigorously quality-checked to ensure compliance and performance.
Global Logistics for Timely Delivery
We collaborate with major courier services like UPS, DHL, and FedEx to ensure timely and safe delivery of your kits. Our logistics team monitors shipments and resolves any issues to guarantee efficient sample collection and transport.
Biospecimen Data and
Patient Data Management

Comprehensive Patient Data
Our advanced technology enables access to comprehensive patient demographics, including age, gender, ethnicity, and more. Researchers can filter data to match biospecimens with specific conditions, treatments, and outcomes, enhancing the precision of your study.
Detailed Testing & Collection Data
Researchers can access robust de-identified patient and specimen data, including testing histories, collection procedures, and test methodologies. This ensures precise matching of biospecimens to research needs.
Storage, Packaging and
Logistics Management

Secure and Controlled Biorepository
SONA offers state-of-the-art biorepository with storage options ranging from ambient temperatures to liquid nitrogen. Our facilities include limited access, redundant backup systems and 24/7 automated temperature monitoring to ensure the highest level of sample safety.
Expert Logistics Management
We manage import/export permits and offer a variety of shipment options—ambient, refrigerated, or frozen. With years of experience working with major couriers, we ensure the safe and timely transport of all samples.
Logistics Support
01.
Support in identifying right logistic provider based on samples need and budget.
02.
Coordinating with the logistics provider to monitor the progress of the shipment
03.
Various storage and logistics options available: LN2, Dry Ice, Ambient Temp.
04.
Special request, such as shipment with data logger, dry ice replenishment, can be facilitated.
05.
Documentation assistance for Import and Export
06.
Global shipments within 48 hours
Quality Assurance
Adherence to SONA Standard Operating Procedures (SOPs)
Adherence to SONA Standard Operating Procedures (SOPs)
-
All biospecimens are collected following SONA’s SOPs, designed in line with ISBER Best Practices and NCI standards
-
Ensures dependable quality metrics for all samples delivered to sponsors
-
Trained personnel oversee specimen collection, processing, deidentification, data recording, storage, and retrieval
-
Preserves sample viability and functionality for research projects
Standardized Processes Across All Sites
Comprehensive Chain of Custody (COC)
Dual Control and Verification Systems
Tissue Quality Control (QC)
Pathologist Dual Review Process
Advanced Analytical Testing (Per Request)
Specimen Integrity Checks
Error-Proof Data Entry and Reporting
The SONA Quality Guarantee
Quality Assurance
Adherence to SONA
(SOPs)
Standardized Processes
Across All Sites
Comprehensive Chain
of Custody (COC)
Dual Control and
Verification Systems
Tissue Quality
Control (QC)
Adherence to SONA
Standard Operating
Procedures (SOPs)
-
All biospecimens are collected following SONA’s SOPs, designed in line with ISBER Best Practices and NCI standards
-
Ensures dependable quality metrics for all samples delivered to sponsors
-
Trained personnel oversee specimen collection, processing, deidentification, data recording, storage, and retrieval
-
Preserves sample viability and functionality for research projects
Pathologist Dual
Review Process
Advanced Analytical
Testing (Per Request)
Specimen
Integrity Checks
Error-Proof Data
Entry and Reporting
The SONA
Quality Guarantee
By leveraging these operational capabilities, SONA Biosciences ensures that your research projects are executed with the highest standards of quality, efficiency, and scientific rigor. Our comprehensive approach to biospecimen management and research support empowers you to focus on your scientific goals while we handle the complexities of logistics, data management, and quality control.
Partner with SONA Biosciences to experience the difference that expert operational capabilities can make in advancing your life science research.
Project Work Flow
At SONA Biosciences, we follow a systematic and efficient project workflow that ensures smooth execution of your research initiatives. Our workflow is designed to maximize efficiency, maintain quality, and adhere to all relevant regulatory standards. The key stages in our project workflow are:
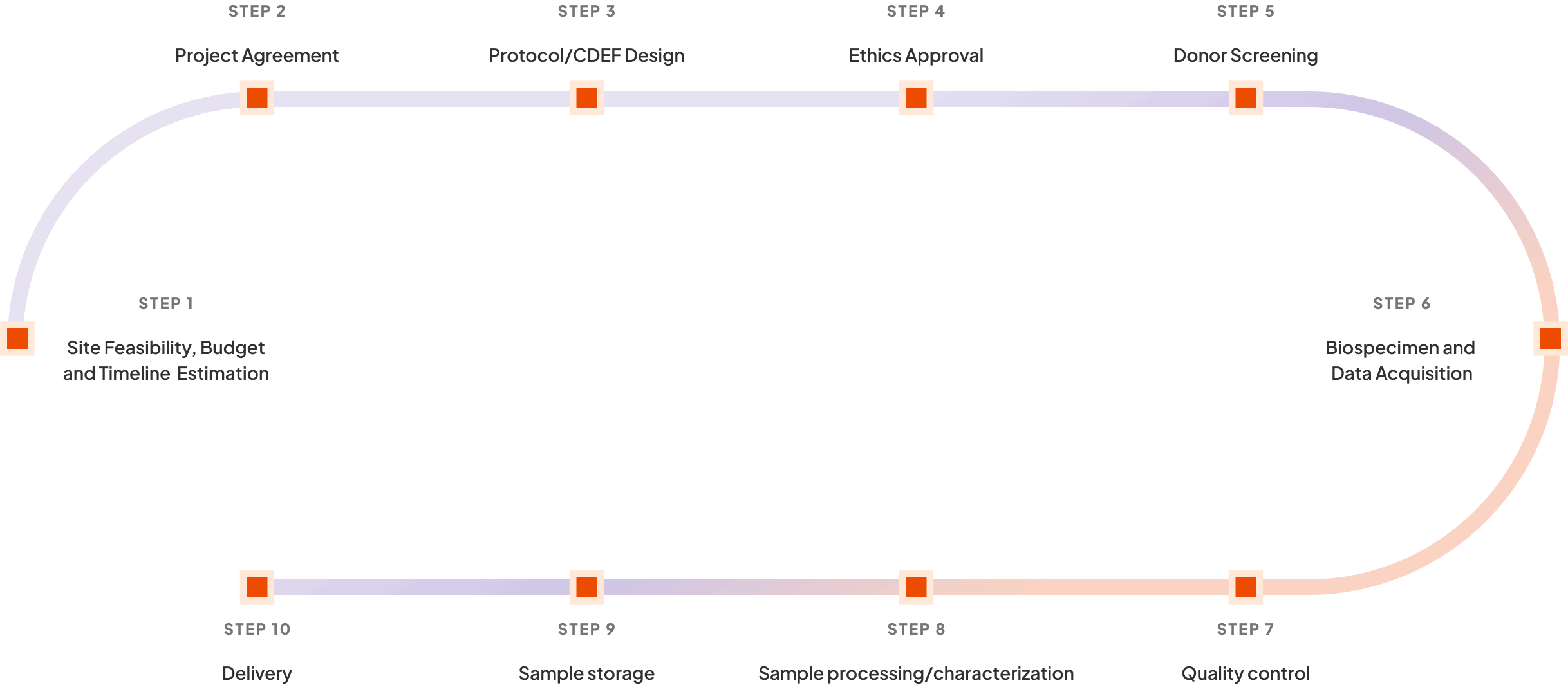
This comprehensive workflow allows us to manage every aspect of your project with precision and care, from initial planning to final delivery.